Alu PCR Lab

Purpose: The purpose of this lab was to analyze our cheek cells DNA to find the
Hypothesis: I think that some people will have the Alu gene and some will not.
Materials: Cheek cells, pipette, Agarose gel, trays, master mix, premier mix, microcentrifuge, TBE Buffer
Procedure:
1. Swirl 10 ml of saline solution in your mouth for 30 seconds. Spit out saline into a cup and swirl to mix cells.
2. Transfer 1000 microliters of the saline/cell suspension into your labeled microfuge tube. Spin in a microcentrifuge to pellet the cells. Pour off the supernatent, allowing 100 microliters to cover the cell pallet. Rack the sample.
3. Withdraw 50 microliters of your cell suspension and add it to a tube containing Chelex.
4. Apply Chelex tube to a heat block for 10 minutes.
5. Remove Chelex tube from heat block. Use a P-200 to withdraw 50 microliters of supernatent from the Chelex tube and transfer to a fresh tube.
6. Obtain a tiny PCR tube and keep on ice.
7. Pipette 20 microliters of Master Mix into the PCR tube. Then add 20 microliters of Primer Mix.
8. Add 10 microliters of your extracted DNa into the PCR tube.
9. Place reaction into a thermal cycler.
10. Retrieve PCR tube and spin in a microcentrifuge. Then, add 5 microliters of loading dye.
11. Create and pour gels. Add 1XTAE solution.
12. Load 15 to 20 microliters of the DNA/loading dye mixture into a well in your gel.
13. Load 5 to 10 microliters of a 100 base-pair ladder (molecular weight marker) into the one well in each gel for later comparison.
14. Run gels.
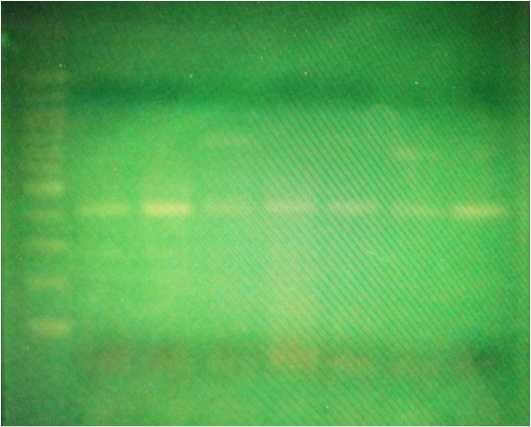
Results: My results shows that i'm +/-, which means i'm a heterozygous.
Analysis/Conclusion: For volumes 1-20 µL, the correct micropipet to use would be P-20. For volumes 20-200 µL, the correct micropipet would P-200. Finally, for volumes 200-1000 µL, the correct micropipet would be P-1000. Both the P-200 and P-1000 can do 100 µL, but the P-200 would work better. Three suggestions I have are to use the smallest micropipet possible for more accuracy, add volumes starting from the smallest and going up to the largest volumes, and making sure to clearly label all of the tubes to ensure you stay organized. I measured my percent of error and it was 15% for the first two trials, and 49% for the last trial. My results were relatively accurate with some errors. Discovering what genotype you have, is an interesting process. This was our first time working with micro pipets which made it a little nerve-racking but also exciting at the same time. I got to learn a little about myself and am looking forward to the other labs in the future.
Hypothesis: I think that some people will have the Alu gene and some will not.
Materials: Cheek cells, pipette, Agarose gel, trays, master mix, premier mix, microcentrifuge, TBE Buffer
Procedure:
1. Swirl 10 ml of saline solution in your mouth for 30 seconds. Spit out saline into a cup and swirl to mix cells.
2. Transfer 1000 microliters of the saline/cell suspension into your labeled microfuge tube. Spin in a microcentrifuge to pellet the cells. Pour off the supernatent, allowing 100 microliters to cover the cell pallet. Rack the sample.
3. Withdraw 50 microliters of your cell suspension and add it to a tube containing Chelex.
4. Apply Chelex tube to a heat block for 10 minutes.
5. Remove Chelex tube from heat block. Use a P-200 to withdraw 50 microliters of supernatent from the Chelex tube and transfer to a fresh tube.
6. Obtain a tiny PCR tube and keep on ice.
7. Pipette 20 microliters of Master Mix into the PCR tube. Then add 20 microliters of Primer Mix.
8. Add 10 microliters of your extracted DNa into the PCR tube.
9. Place reaction into a thermal cycler.
10. Retrieve PCR tube and spin in a microcentrifuge. Then, add 5 microliters of loading dye.
11. Create and pour gels. Add 1XTAE solution.
12. Load 15 to 20 microliters of the DNA/loading dye mixture into a well in your gel.
13. Load 5 to 10 microliters of a 100 base-pair ladder (molecular weight marker) into the one well in each gel for later comparison.
14. Run gels.
Results: My results shows that i'm +/-, which means i'm a heterozygous.
Analysis/Conclusion: For volumes 1-20 µL, the correct micropipet to use would be P-20. For volumes 20-200 µL, the correct micropipet would P-200. Finally, for volumes 200-1000 µL, the correct micropipet would be P-1000. Both the P-200 and P-1000 can do 100 µL, but the P-200 would work better. Three suggestions I have are to use the smallest micropipet possible for more accuracy, add volumes starting from the smallest and going up to the largest volumes, and making sure to clearly label all of the tubes to ensure you stay organized. I measured my percent of error and it was 15% for the first two trials, and 49% for the last trial. My results were relatively accurate with some errors. Discovering what genotype you have, is an interesting process. This was our first time working with micro pipets which made it a little nerve-racking but also exciting at the same time. I got to learn a little about myself and am looking forward to the other labs in the future.